Environmental Effects of Coal Burning
10:56 AM
Posted by Energetic
There are a number of adverse health and environmental effects of coal burning especially in power stations, and of coal mining. These effects include:
- Coal-fired power plants shorten nearly 24,000 lives a year in the United States, including 2,800 from lung cancer
- Generation of hundreds of millions of tons of waste products, including fly ash, bottom ash, flue gas desulfurization sludge, that contain mercury, uranium, thorium, arsenic, and other heavy metals
- Acid rain from high sulfur coal
- Interference with groundwater and water table levels
- Contamination of land and waterways and destruction of homes from fly ash spills such as Kingston Fossil Plant coal fly ash slurry spill
- Impact of water use on flows of rivers and consequential impact on other land-uses
- Dust nuisance
- Subsidence above tunnels, sometimes damaging infrastructure
- Uncontrollable underground fires which may burn for decades or centuries.
- Coal-fired power plants without effective fly ash capture are one of the largest sources of human-caused background radiation exposure
- Coal-fired power plants emit mercury, selenium, and arsenic which are harmful to human health and the environment
- Release of carbon dioxide, a greenhouse gas, which causes climate change and global warming according to the IPCC and the EPA. Coal is the largest contributor to the human-made increase of CO2 in the air
Carbon Intensity of Coal
7:27 AM
Posted by Energetic
Commercial coal has a carbon content of at least 70%. Coal with a heating value of 6.67 kWh per kilogram as quoted above has a carbon content of roughly 80%, which is
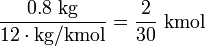 , where 1 mol equals to NA (Avogadro Number) atoms.
, where 1 mol equals to NA (Avogadro Number) atoms.
Carbon combines with oxygen in the atmosphere during combustion, producing carbon dioxide, with an atomic weight of (12 + 16 × 2 = 44 kg/kmol). The CO2 released to air for each kilogram of incinerated coal is therefore
 .
.
This can be used to calculate an emission factor for CO2 from the use of coal power. Since the useful energy output of coal is about 30% of the 6.67 kWh/kg(coal), the burning of 1 kg of coal produces about 2 kWh of electrical energy. Since 1 kg coal emits 2.93 kg CO2, the direct CO2 emissions from coal power are 1.47 kg/kWh, or about 0.407 kg/MJ.
The U.S. Energy Information Agency's 1999 report on CO2 emissions for energy generation, quotes a lower emission factor of 0.963 kg CO2/kWh for coal power. The same source gives a factor for oil power in the U.S. of 0.881 kg CO2/kWh, while natural gas has 0.569 kg CO2/kWh. Estimates for specific emission from nuclear power, hydro, and wind energy vary, but are about 100 times lower.
Energy Density of Coal
7:26 AM
Posted by Energetic
The energy density of coal, i.e. its heating value, is roughly 24 megajoules per kilogram.
The energy density of coal can also be expressed in kilowatt-hours, the units that electricity is most commonly sold in, per units of mass to estimate how much coal is required to power electrical appliances. One kilowatt-hour is 3.6 MJ, so the energy density of coal is 6.67 kW·h/kg. The typical thermodynamic efficiency of coal power plants is about 30%, so of the 6.67 kW·h of energy per kilogram of coal, 30% of that—2.0 kW·h/kg—can successfully be turned into electricity; the rest is waste heat. So coal power plants obtain approximately 2.0 kW·h per kilogram of burned coal.
As an example, running one 100-watt lightbulb for one year requires 876 kW·h (100 W × 24 h/day × 365 day/year = 876000 W·h = 876 kW·h). Converting this power usage into physical coal consumption:
It takes 325 kg (714 lb) of coal to power a 100 W lightbulb for one year. One should also take into account transmission and distribution losses caused by resistance and heating in the power lines, which is in the order of 5–10%, depending on distance from the power station and other factors.
Refined Coal
7:03 AM
Posted by Energetic
Commercial Development of Refined Coal
United States
Evergreen Energy constructed a full-scale coal refinery near Gillette, Wyoming that began operation in late 2005. Designed originally to be a commercial plant, the facility encountered design and operational problems. Evergreen idled the facility in March 2008 and instead used the plant as a process development platform with its engineering, construction and procurement contractor Bechtel Power Corporation.
Evergreen is now seeking to construct a coal refinery using the improved Bechtel design at locations in the Midwestern United States and in Asia.
Australia
Calleja Group constructed a full-scale 16,000 tonne per annum pilot demonstration plant at JBD Business Park at Maddingley Mine near Bacchus Marsh, Victoria that began operation in early 2004. From 2005 ECT Limited upgraded the facility, added a water recovery process with Victorian Government funding in 2007 and operated the plant as a process development platform with its engineering partner ARUP. In 2009 ECT Limited secured and agreement with Thang Long Investment Company (Tincom) of Vietnam to finalise commercial feasibility ahead of construction of a 2 million tonne pa export plant by 2014 and 20 million tonne pa export by 2020. ECT Limited is using the ARUP improved design to secure technology licensing agreements with brown coal suppliers in China, India, Indonesia, Poland, Greece and Russia.
Coal Liquefaction Methods
8:39 AM
Posted by Energetic
The Coal liquefaction processes are classified as direct conversion to liquids processes and indirect conversion to liquids processeses. Direct processes are carbonization and hydrogenation.
Pyrolysis and carbonization processes
There are a number of carbonization processes. The carbonization conversion occurs through pyrolysis or destructive distillation, and it produces condensable coal tar, oil and water vapor, non-condensable synthetic gas, and a solid residue-char. The condensed coal tar and oil are then further processed by hydrogenation to remove sulfur and nitrogen species, after which they are processed into fuels.
The typical example of carbonization is the Karrick process. The process was invented by Lewis Cass Karrick in the 1920s. The Karrick process is a low-temperature carbonization process, where coal is heated at 680 °F (360 °C) to 1,380 °F (750 °C) in the absence of air. These temperatures optimize the production of coal tars richer in lighter hydrocarbons than normal coal tar. However, the produced liquids are mostly a by-product and the main product is semi-coke, a solid and smokeless fuel.
The COED Process, developed by FMC Corporation, uses a fluidized bed for processing, in combination with increasing temperature, through four stages of pyrolysis. Heat is transferred by hot gases produced by combustion of part of the produced char. A modification of this process, the COGAS Process, involves the addition of gasification of char. The TOSCOAL Process, an analogue to the TOSCO II oil shale retorting process and Lurgi-Ruhrgas process, which is also used for the shale oil extraction, uses hot recycled solids for the heat transfer.
Liquid yields of pyrolysis and Karrick processes are generally low for practical use for synthetic liquid fuel production. Furthermore, the resulting liquids are of low quality and require further treatment before they can be used as motor fuels. In summary, there is little possibility that this process will yield economically viable volumes of liquid fuel.
Hydrogenation processes
One of the main methods of direct conversion of coal to liquids by hydrogenation process is the Bergius process. The Bergius process was developed by Friedrich Bergius in 1913. In this process, dry coal is mixed with heavy oil recycled from the process. Catalyst is typically added to the mixture. The reaction occurs at between 400 °C (752 °F) to 5,000 °C (9,030 °F) and 20 to 70 MPa hydrogen pressure. The reaction can be summarized as follows:
- n C + (n + 1) H2 → CnH2 n + 2
After World War I several plants were built in Germany; these plants were extensively used during World War II to supply Germany with fuel and lubricants. The Kohleoel Process, developed in Germany by Ruhrkohle and VEBA, was used in the demonstration plant with the capacity of 200 ton of lignite per day, built in Bottrop, Germany. This plant operated from 1981 to 1987. In this process, coal is mixed with a recycle solvent and iron catalyst. After preheating and pressurizing, H2 is added. The process takes place in a tubular reactor at the pressure of 300 bar and at the temperature of 470 °C (880 °F). This process was also explored by SASOL in South Africa.
In 1970-1980s, Japanese companies Nippon Kokan, Sumitomo Metal Industries and Mitsubishi Heavy Industries developed the NEDOL process. In this process, coal is mixed with a recycled solvent and a synthetic iron-based catalyst; after preheating H2 is added. The reaction takes place in a tubular reactor at temperature between 430 °C (810 °F) and 465 °C (870 °F) at the pressure 150-200 bar. The produced oil has low quality and requires intensive upgrading. H-Coal process, developed by Hydrocarbon Research, Inc., in 1963, mixes pulverized coal with recycled liquids, hydrogen and catalyst in the ebullated bed reactor. Advantages of this process are that dissolution and oil upgrading are taking place in the single reactor, products have high H/C ratio, and a fast reaction time, while the main disadvantages are high gas yield (this is basically a thermal cracking process), high hydrogen consumption, and limitation of oil usage only as a boiler oil because of impurities.
The SRC-I and SRC-II (Solvent Refined Coal) processes developed by Gulf Oil and implemented as pilot plants in the United States in the 1960s and 1970s. The Nuclear Utility Services Corporation developed hydrogenation process which was patented by Wilburn C. Schroeder in 1976. The process involved dried, pulverized coal mixed with roughly 1wt% molybdenum catalysts. Hydrogenation occurred by use of high temperature and pressure synthesis gas produced in a separate gasifier. The process ultimately yielded a synthetic crude product, Naphtha, a limited amount of C3/C4 gas, light-medium weight liquids (C5-C10) suitable for use as fuels, small amounts of NH3 and significant amounts of CO2. Other single-stage hydrogenation processes are the Exxon Donor Solvent Process, the Imhausen High-pressure Process, and the Conoco Zinc Chloride Process.
There is also a number of two-stage direct liquefaction processes; however, after 1980s only the Catalytic Two-stage Liquefaction Process, modified from the H-Coal Process; the Liquid Solvent Extraction Process by British Coal; and the Brown Coal Liquefaction Process of Japan have been developed.
Shenhua, the Chinese coal miner, decided in 2002 to build a direct liquefaction plant in Inner Mongolia, with barrel capacity of 20 thousand barrels per day (3.2×103 m3/d). First tests were implemented at the end of 2008. A second and longer test campaign was started in October 2009.
Chevron Corporation developed a process invented by Joel W. Rosenthal called the Chevron Coal Liquefaction Process (CCLP). It is unique due the close-coupling of the non-catalytic dissolver and the catalytic hydroprocessing unit. The oil produced had properties that were unique when compared to other coal oils; it was lighter and had far fewer heteroatom impurities. The process was scaled-up to the 6 ton per day level, but not proven commercially.
Indirect conversion processes
The main indirect process is the Fischer-Tropsch process. In this process, coal is first gasified to make syngas (a balanced purified mixture of CO and H2 gas). Next, Fischer-Tropsch catalysts are used to convert the syngas into light hydrocarbons (like ethane) which are further processed into gasoline and diesel. This method was used on a large technical scale in Germany between 1934 and 1945 and is currently being used by Sasol in South Africa. In addition to creating gasoline, syngas can also be converted into methanol, which can be used as a fuel, or into a fuel additive.
Syngas may be converted to liquids through conversion of the syngas to methanol which is subsequently polymerized into alkanes over a zeolite catalyst. This process, named as the Mobil MTG Process, was developed by Mobil in early 1970s.
Environmental Effects of Coal Gasification
6:01 AM
Posted by Energetic
In the earliest days of MGP operations, coal tar was considered a waste and often disposed into the environment in and around the plant locations. While uses for coal tar developed by the late-19th century, the market for tar varied and plants that could not sell tar at a given time could store tar for future use, attempt to burn it as fuel for the boilers, or dump the tar as waste. Commonly, waste tars were disposed of in old gas holders, adits or even mine shafts (if present). Over time, the waste tars degrade with phenols, benzene (and other mono-aromatics – BTEX) and polycyclic aromatic hydrocarbons released as pollutant plumes that can escape into the surrounding environment. Other wastes included "blue billy", which is a ferroferricyanide compound—the blue colour is from Prussian blue, which was commercially used as a dye. Blue billy is typically a granular material and was sometimes sold locally with the strap line "guaranteed weed free drives". The presence of blue billy can give gas works waste a characteristic musty/bitter almonds or marzipan smell which is associated with cyanide gas.
The shift to the CWG process initially resulted in a reduced output of water gas tar as compared to the volume of coal tars. The advent of automobiles reduced the availability of naphtha for carburetion oil, as that fraction was desirable as motor fuel. MGPs that shifted to heavier grades of oil often experienced problems with the production of tar-water emulsions, which were difficult, time consuming, and costly to break. (The cause of tar-water emulsions is complex and was related to several factors, including free carbon in the carburetion oil and the substitution of bituminous coal as a feedstock instead of coke.) The production of large volumes of tar-water emulsions quickly filled up available storage capacity at MGPs and plant management often dumped the emulsions in pits, from which they may or may not have been later reclaimed. Even if the emulsions were reclaimed, the environmental damage from placing tars in unlined pits remained. The dumping of emulsions (and other tarry residues such as tar sludges, tank bottoms, and off-spec tars) into the soil and waters around MGPs is a significant factor in the pollution found at FMGPs today.
Commonly associated with former manufactured gas plants (known as "FMGPs" in environmental remediation) are contaminants including:
- BTEX
- Diffused out from deposits of coal/gas tars
- Leaks of carburetting oil/light oil
- Leaks from drip pots, that collected condensible hydrocarbons from the gas
- Coal tar waste/sludge
- Typically found in sumps of gas holders/decanting ponds.
- Coal tar sludge has no resale value and so was always dumped.
- Volatile organic compounds
- Polycyclic aromatic hydrocarbons (PAHs)
- Present in coal tar, gas tar, and pitch at significant concentrations.
- Heavy metals
- Leaded solder for gas mains, lead piping, coal ashes.
- Cyanide
- Purifier waste has large amounts of complex ferrocyanides in it.
- Lampblack
- Only found where crude oil was used as gasification feedstock.
- Tar emulsions
Coal tar and coal tar sludges are frequently denser than water and are present in the environment as a dense non-aqueous phase liquid.
In the UK, former gasworks have commonly been developed over for residential and other uses (including the Millennium Dome), being seen as prime developable land in the confines of city boundaries. Situations such as these are now lead to problems associated with planning and the Contaminated Land Regime and have recently been debated in the House of Commons.
The more modern coal gasification processes (circa 1970 to 2006) also have environmental problems requiring various available technologies for mitigation.UCG Environmental and Social Impacts
10:08 AM
Posted by Energetic
Eliminating mining eliminates mine safety issues. Compared to traditional coal mining and processing the underground coal gasification eliminates surface damage and solid waste discharge, and reduces sulfur dioxide (SO2) and nitrogen oxide (NOx) emissions. For comparison, the ash content of UCG syngas is estimated to be approximately 10 mg/m³ compared to smoke from traditional coal burning where ash content may be up to 70 mg/m³. However, UCG operations cannot be controlled as precisely as surface gasifiers. Variable include the rate of water influx, the distribution of reactants in the gasification zone, and the growth rate of the cavity. These can only be estimated from temperature measurements, and analyzing product gas quality and quantity.
Subsidence is a common issue with all forms of extractive industry. While UCG leaves the ash behind in the cavity, the depth of the void left after UCG is typically more than other methods of coal extraction.
Underground combustion produces NOx and SO2 and lowers emissions, including acid rain. The process has advantages for geologic carbon storage. Combining UCG with CCS technology allows re-injecting some of the CO2 on-site into the highly permeable rock created during the burning process, i.e. where the coal used to be. Contaminants such as ammonia and hydrogen sulfide can be removed from product gas at a relatively low cost.
Aquifer contamination is a potential environmental concerns. Organic and often toxic materials (such as phenol) remain in the underground chamber after gasification and therefore are likely to leach into ground water, absent appropriate site selection. Phenol leachate is the most significant environmental hazard due to its high water solubility and high reactiveness to gasification. Livermore conducted a burn at Hoe Creek, Wyoming, producing operating pressure in the burn cavity greater than the surrounding rock, forcing contaminants (including the carcinogen benzene) into potable groundwater. However, some research has shown that the persistence of such substances in the water is short and that ground water recovers within two yearsUnderground Coal Gasification
10:03 AM
Posted by Energetic

Process of Underground Coal Gasification
Underground coal gasification converts coal to gas while still in the coal seam (in-situ). Gas is produced and extracted through wells drilled into the unmined coal–seam. Injection wells are used to supply the oxidants (air, oxygen, or steam) to ignite and fuel the underground combustion process. Separate production wells are used to bring the product gas to surface. The high pressure combustion is conducted at temperature of 700–900 °C (1290–1650 °F), but it may reach up to 1,500 °C (2,730 °F). The process decomposes coal and generates carbon dioxide (CO2), hydrogen (ḥ), carbon monoxide (CO) and small quantities of methane (CH4) and hydrogen sulfide(H2S). As the coal face burns and the immediate area is depleted, the oxidants injected are controlled by the operator.
As coal varies considerably in its resistance to flow, depending on its age, composition and geological history, the natural permeability of the coal to transport the gas is generally not adequate. For high pressure break-up of the coal, hydro-fracturing, electric-linkage, and reverse combustion may be used in varying degrees.
Two methods are commercially available. One uses vertical wells and a method of reverse combustion to open internal pathways in the coal. The process was used in the Soviet Union and was later modified by Ergo Exergy. It was tested in Chinchilla site in 1998–2003. Livermore developed another method that creates dedicated inseam boreholes, using drilling and completion technology adapted from oil and gas production. It has a movable injection point known as CRIP (controlled retraction injection point) and generally uses oxygen or enriched air for gasification.
According to the Commonwealth Scientific and Industrial Research Organisation the following coal seam characteristics are most suitable for the underground coal gasification:
- Depth of 100–600 metres (330–2,000 ft)
- Thickness more than 5 metres (16 ft)
- Ash content less than 60%
- Minimal discontinuities
- No nearby aquifers (to avoid polluting supplies of drinking water).
